Compliance ERP vs. Legacy Systems in the Chemical Industry
A decade ago, many chemical manufacturers viewed ERP as record-keeping, but today compliance ERP and chemical compliance software have become central engines for operational safety. In 2025, it’s a central compliance engine, a connected ecosystem, that ties OSHA Hazard Communication (HazCom) requirements, GHS labeling, safety training, and access control into a single, real-time platform.
The stakes are higher than ever. Penalties for willful violations have more than doubled in some states. Regulators expect faster evidence retrieval, live process traceability, and role-based clarity in hazardous material handling. The companies that succeed are the ones that stop treating compliance as a file cabinet function and start treating it as an operational performance metric.
From SDS Compliance to Real-Time Hazard Intelligence
Unlike traditional SDS management, ERP-driven chemical data management systems update sheets automatically, ensuring version control and compliance consistency. Today’s ERP-driven SDS workflows do more:
Dynamic Updates
An ingredient change in one formulation triggers automatic SDS and label updates across all affected SKUs, with instant downstream version control.
Multi-Channel Access
QR codes on labels pull the latest SDS directly from the ERP, whether scanned on the plant floor, in transit, or at a customer site.
Role-Based Gatekeeping
Access to hazardous substances is automatically restricted to certified, trained staff. The ERP cross-checks training validity in real time via LMS integration before issuing approval.
This isn’t theoretical, in one chemical plant retrieval time for SDS during a regulatory inspection dropped from 25 minutes to under 60 seconds after ERP integration. That alone can mean the difference between passing and failing an audit.
Building a Connected Chemical Compliance System
Modern chemical compliance systems, powered by ERP and compliance management software, go beyond individual features to provide an integrated ecosystem. By integrating SDS, training, labeling, and audit readiness into a seamlessly connected ecosystem, they transform compliance from a post-operational task into an integral part of the production process.
Instead of siloed systems, ERP-driven ecosystems provide a real-time digital safety net—ensuring that every update, approval, and access check reinforces operational safety and regulatory alignment across the enterprise.
ERP + MES + GenAI = Predictive Compliance
Forward-looking chemical manufacturers are linking ERP and MES to create compliance-ready production environments. By adding GenAI-driven insights, systems can now:
- Flag training gaps before a scheduled audit
- Suggest preventive maintenance schedules for equipment tied to hazardous processes
- Integrated PPE ERP capabilities recommend updated protective equipment protocols when OSHA or GHS standards shift, closing gaps in worker safety compliance
- Generate “risk readiness dashboards” so leadership can see immediately where exposure exists before the regulator does
One chemical distributor we worked with used this combined approach to cut OSHA corrective action timelines from 21 days to just 6.
Why ERP Security Matters in the Chemical Industry
ERP security in the chemical industry ensures that hazardous data, SDS records, and compliance workflows are protected across region-specific regulatory environments. An ERP that isn’t configured for SDS, HazCom, and GHS workflows from the ground up creates hidden risk:
- Incomplete hazard labeling for export shipments
- Mismatched SDS data between facilities
- Missed training renewals for high-risk roles
Modern compliance ERP solutions ensure GHS labeling accuracy with ERP integration, where every formula change instantly updates hazard classifications and SDS sheets across facilities.
Every formula change instantly updates hazard classifications, pictograms, and SDS sheets across facilities, eliminating manual rework and ensuring global consistency. Automated version control ensures that the latest SDS is always available, while integration with labeling systems reduces the chance of costly misprints or outdated hazard symbols.
Working with a vendor who understands chemical manufacturing means your ERP is built to speak compliance fluently, not just store it.
ERP Compliance for Chemical Industry OSHA Standards
The conversation has shifted from “How do we avoid fines?” to “How do we use compliance as proof of operational excellence?”
- Investors see it as a proxy for governance quality
- Customers see it as a trust marker
- Regulators see it as an indicator of cultural maturity
An ERP built for compliance in the chemical industry not only tracks OSHA standards and CAPA readiness, but also turns audit prep into a source of operational credibility.
Bottom Line
The era of treating ERP as a compliance archive has ended. In the modern chemical industry, managing compliance is no longer a reactive burden but a strategic imperative. A purpose-built ERP system now acts as the central nervous system of operations, seamlessly orchestrating people, processes, and data into a connected ecosystem that delivers real-time safety intelligence.
This transformation makes regulatory adherence a built-in operational reflex: OSHA inspections, automated GHS labeling, SDS updates, hazardous material tracking. By shifting from manual, after-the-fact checks to automated, ecosystem-wide visibility, chemical manufacturers don’t just respond faster – they stay audit-ready year-round.
Most importantly, compliance stops being a cost of doing business and becomes a source of sustained competitive advantage and profitability. With the right ERP foundation, risk readiness isn’t just protection – it’s performance.
Don’t wait for audits to expose risks – ERP for chemical compliance keeps you OSHA- and GHS-ready every day.
At a Glance
- The Pharmaceutical industry has witnessed exponential growth during the past decade, and pharma revenues worldwide reached approximately 1.6 trillion U.S. dollars in 2023, growing to an estimated 1.77 trillion U.S. dollars in 2024 – Per Statista.
- This growth rate automatically created a need to boost supply chain visibility and inventory traceability.
- Regulatory measures put forth by the government or agencies such as the FDA can create unforeseen challenges for Pharma companies.
- Leveraging technology’s power and identifying solutions for Pharma to streamline operations can effectively boost your bottom-line and help in better quality control while meeting both regulations and market demands together.
Understanding the Pain Points
The inability to adapt to technological advancements and rapidly changing regulatory landscape makes managing pharmaceutical supply chains quite a tough pill to swallow. Successful pharmaceutical companies leverage the power of IT to streamline their supply chain operations.
Figure 1:Pain Points in Pharmaceutical Industry

The pharma industry has established itself as one of the fastest-growing industries in the past few decades, with a significant share of its investments in research, marketing, manufacturing, and development of its products.
In the present scenario, the table seems to be turning. The industry is struggling to keep pace with rapidly changing regulations and dealing with challenges such as:
- Keeping up with the growing competition due to faster technological advances
- The rapid development of new products to handle growing health concerns.
- Healthcare providers and government reforms put downward pressure on product prices of the industry.
- Full traceability of the product pipeline to be on par with requirements like quality compliance, serialization mandate, and the rise of counterfeited drugs.
- Issues related to delivering products to the market.
The above challenges highlight the importance of comprehensive solutions that help Pharma companies progress and overcome various hurdles in shipping products to customers.
Collaboration Within the Pharma Value Chain
To keep up with the growing list of challenges, Pharma companies are increasingly relying on third parties like CROs, CMOs, 3PLs to manage the following:
- Outsource research & development.
- Manufacturing of intermediates.
- 3rd party logistics for warehouse and inventory management.
- Quality control testing.
- Shipping and distribution of products.
While this is beneficial for pharmaceutical companies, it comes with its complexities, including the remote location of different departments, inventory, and testers. How do you run a pharmaceutical supply chain effectively when all the departments are functioning from other locations? How would you effectively manage workflows that involve the exchange of information with outsourced parties on multiple tiers of your supply?
As of 2024, the United States faced an all-time high of over 300 different kinds of drug shortages in healthcare and pharmacy settings. A 2024 report showed that 62% of all drug shortages were caused by manufacturing and product quality problems resulting in supply disruptions.
Following are a few benefits for Pharma companies to be technology-centric:
- Real-time & end-to-end supply chain visibility.
- Collaboration with the Pharma value chain.
- Optimized inventories with least stock out situations.
- Higher margins through smarter channel allocations for an increasing share in the market.
- Improved customer experience.
- Visual analytics and reporting.
Digitize the Pharma Supply Chain
Companies require well-streamlined processes and collaboration using agile software solutions for Pharma that provide significant cost savings and flexibility. Technology for the Pharmaceutical industry can be a helping hand to optimize the supply chains. Advanced Solutions for Pharma increase inventory visibility and lot traceability across your supply chain, ultimately improving the overall product quality. Post COVID-19, embracing technology as a growth enabler, has become even more critical, with 89% of pharma industry leaders implementing or developing digital transformation strategies as of 2024.
Following the path of other industries like hi-tech and consumer goods, the Pharma companies also need to focus on these five initiatives:
01. Connecting and collaborating using a working business network
A digital business network sets the foundation of a multi-enterprise supply chain, where all departments are interconnected on the cloud, ensuring seamless information flow and collaboration. Unlike the outdated model based on point-to-point connections, this multi-tier network is much advanced and helps end-users stay in sync with counterparts and move quickly through the process. The ability to collaborate enables end-to-end visibility and collaboration – the two critical factors that support smooth business interactions among different business partners involved in the value chain. Seamless integration helps you get real-time visibility and coordination in your entire extended supply chain, including multiple partners.
Figure 2:Process of Temperature Control
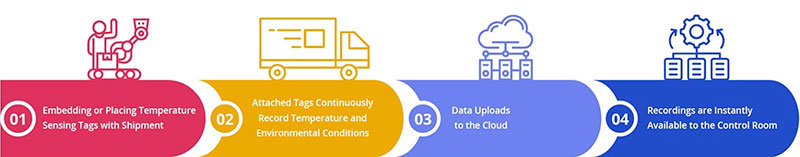
For example, there can be immense damage caused to vaccines due to the drop in temperature in the storage areas while being transported from one place to another. Real-time data tracking can help with adequate temperature controls by notifying people when the temperatures are above or below the desired levels.
02. Estimating true demand
Demand predictions are just well-painted guesses of how much of a percentage of the population would require a particular product in the future. Most innovative pharmaceutical companies are now following demand forecasting as per consumer product companies for their OTC products. They do this by capturing massive volumes of demand-related data and feeding it into sophisticated demand planning software, which helps them predict demand effectively. The data or demand forecasts captured are shared with all supply chain partners, ensuring the pharma companies, suppliers, and CMOs align with the arrangement. Estimating actual demand helps them to bring significantly higher on-shelf availability and lowers inventory.
03. Control quality for CMOs
Pharmaceutical companies need to ensure end-to-end traceability. How do they keep track of their pharmaceutical manufacturing supply chain when a CMO is involved? Having visibility into partner manufacturing operations and tracking product quality across the multi-tier system is essential for pharma companies. Real-time visibility can be done by connecting CMOs to your Manufacturing Execution Systems. A manufacturing execution system helps capture relevant data at all production stages, providing granular visibility into plant operations. Pharma companies will effectively track material flows, lot of genealogy, processing steps, and associated parameters, such as yields or test results – critical information to ensure traceability. Quality control is of optimal importance for any serialization initiative within pharmaceutical companies.
04. Faster planning across the network
Another essential requirement for companies is to detect and respond to changes in supply and demand faster. With largely connected systems and expanding business networks, companies have a clear, end-to-end visibility of the supply chain in the Pharma company and not just in-house operations; this includes expected lead times on inventory from a contract manufacturer or raw material supplier. However, traditional planning systems can impact your decision-making capabilities required to manage inventory shortages and suggest alternative purchase/ manufacturing scenarios. Advanced planning tools allow rapid evaluation of new buy-sell signals and easily recommend purchases from secondary vendors or alternate manufacturing routes. This new plan can then be shared with all the supply chain partners within the value chain through collaboration portals, tools, and so forth.
05. Manage distribution
Pharma companies are increasingly relying on third parties for transportation, warehousing, and other value-added services. Ensuring product availability implies proper management of distribution partners. For downstream visibility, inventory management coupled with sophisticated stock strategies are needed. Proactive pharma supply chain management helps all products be delivered to the right customers to increase your sell-metric, which is critical when competing in the market.
Pharma companies are customizing supply chains to suit the product types being transported, with digitally mature pharma companies reducing development timelines by up to 30% through the implementation of real-time visibility, IoT sensors, and blockchain technology for enhanced transparency and security.
Key Takeaways
- The expectation of better customer experience is the same across all industries and progressive Lifesciences companies understand the need to embrace digital for Pharma.
- More Pharma companies are continually evaluating ways to address supply chain challenges to meet customer needs ultimately.
- A cloud-based agile technology network to increase visibility in the supply chain and keep all functions connected for better collaboration can boost your profits and increase growth potential.
See pharmaceutical supply chain optimization in action.
Understanding the Pain Points
The inability to adapt to technological advancements and rapidly changing regulatory landscape makes managing pharmaceutical supply chains quite a tough pill to swallow. Successful pharmaceutical companies leverage the power of IT to streamline their supply chain operations.
Figure 1:Pain Points in Pharmaceutical Industry

The pharma industry has established itself as one of the fastest-growing industries in the past few decades, with a significant share of its investments in research, marketing, manufacturing, and development of its products.
In the present scenario, the table seems to be turning. The industry is struggling to keep pace with rapidly changing regulations and dealing with challenges such as:
- Keeping up with the growing competition due to faster technological advances
- The rapid development of new products to handle growing health concerns.
- Healthcare providers and government reforms put downward pressure on product prices of the industry.
- Full traceability of the product pipeline to be on par with requirements like quality compliance, serialization mandate, and the rise of counterfeited drugs.
- Issues related to delivering products to the market.
The above challenges highlight the importance of comprehensive solutions that help Pharma companies progress and overcome various hurdles in shipping products to customers.
Collaboration Within the Pharma Value Chain
To keep up with the growing list of challenges, Pharma companies are increasingly relying on third parties like CROs, CMOs, 3PLs to manage the following:
- Outsource research & development.
- Manufacturing of intermediates.
- 3rd party logistics for warehouse and inventory management.
- Quality control testing.
- Shipping and distribution of products.
While this is beneficial for pharmaceutical companies, it comes with its complexities, including the remote location of different departments, inventory, and testers. How do you run a pharmaceutical supply chain effectively when all the departments are functioning from other locations? How would you effectively manage workflows that involve the exchange of information with outsourced parties on multiple tiers of your supply?
As of 2024, the United States faced an all-time high of over 300 different kinds of drug shortages in healthcare and pharmacy settings. A 2024 report showed that 62% of all drug shortages were caused by manufacturing and product quality problems resulting in supply disruptions.
Following are a few benefits for Pharma companies to be technology-centric:
- Real-time & end-to-end supply chain visibility.
- Collaboration with the Pharma value chain.
- Optimized inventories with least stock out situations.
- Higher margins through smarter channel allocations for an increasing share in the market.
- Improved customer experience.
- Visual analytics and reporting.
Digitize the Pharma Supply Chain
Companies require well-streamlined processes and collaboration using agile software solutions for Pharma that provide significant cost savings and flexibility. Technology for the Pharmaceutical industry can be a helping hand to optimize the supply chains. Advanced Solutions for Pharma increase inventory visibility and lot traceability across your supply chain, ultimately improving the overall product quality. Post COVID-19, embracing technology as a growth enabler, has become even more critical, with 89% of pharma industry leaders implementing or developing digital transformation strategies as of 2024.
Following the path of other industries like hi-tech and consumer goods, the Pharma companies also need to focus on these five initiatives:
01. Connecting and collaborating using a working business network
A digital business network sets the foundation of a multi-enterprise supply chain, where all departments are interconnected on the cloud, ensuring seamless information flow and collaboration. Unlike the outdated model based on point-to-point connections, this multi-tier network is much advanced and helps end-users stay in sync with counterparts and move quickly through the process. The ability to collaborate enables end-to-end visibility and collaboration – the two critical factors that support smooth business interactions among different business partners involved in the value chain. Seamless integration helps you get real-time visibility and coordination in your entire extended supply chain, including multiple partners.
Figure 2:Process of Temperature Control

For example, there can be immense damage caused to vaccines due to the drop in temperature in the storage areas while being transported from one place to another. Real-time data tracking can help with adequate temperature controls by notifying people when the temperatures are above or below the desired levels.
02. Estimating true demand
Demand predictions are just well-painted guesses of how much of a percentage of the population would require a particular product in the future. Most innovative pharmaceutical companies are now following demand forecasting as per consumer product companies for their OTC products. They do this by capturing massive volumes of demand-related data and feeding it into sophisticated demand planning software, which helps them predict demand effectively. The data or demand forecasts captured are shared with all supply chain partners, ensuring the pharma companies, suppliers, and CMOs align with the arrangement. Estimating actual demand helps them to bring significantly higher on-shelf availability and lowers inventory.
03. Control quality for CMOs
Pharmaceutical companies need to ensure end-to-end traceability. How do they keep track of their pharmaceutical manufacturing supply chain when a CMO is involved? Having visibility into partner manufacturing operations and tracking product quality across the multi-tier system is essential for pharma companies. Real-time visibility can be done by connecting CMOs to your Manufacturing Execution Systems. A manufacturing execution system helps capture relevant data at all production stages, providing granular visibility into plant operations. Pharma companies will effectively track material flows, lot of genealogy, processing steps, and associated parameters, such as yields or test results – critical information to ensure traceability. Quality control is of optimal importance for any serialization initiative within pharmaceutical companies.
04. Faster planning across the network
Another essential requirement for companies is to detect and respond to changes in supply and demand faster. With largely connected systems and expanding business networks, companies have a clear, end-to-end visibility of the supply chain in the Pharma company and not just in-house operations; this includes expected lead times on inventory from a contract manufacturer or raw material supplier. However, traditional planning systems can impact your decision-making capabilities required to manage inventory shortages and suggest alternative purchase/ manufacturing scenarios. Advanced planning tools allow rapid evaluation of new buy-sell signals and easily recommend purchases from secondary vendors or alternate manufacturing routes. This new plan can then be shared with all the supply chain partners within the value chain through collaboration portals, tools, and so forth.
05. Manage distribution
Pharma companies are increasingly relying on third parties for transportation, warehousing, and other value-added services. Ensuring product availability implies proper management of distribution partners. For downstream visibility, inventory management coupled with sophisticated stock strategies are needed. Proactive pharma supply chain management helps all products be delivered to the right customers to increase your sell-metric, which is critical when competing in the market.
Pharma companies are customizing supply chains to suit the product types being transported, with digitally mature pharma companies reducing development timelines by up to 30% through the implementation of real-time visibility, IoT sensors, and blockchain technology for enhanced transparency and security.
Key Takeaways
- The expectation of better customer experience is the same across all industries and progressive Lifesciences companies understand the need to embrace digital for Pharma.
- More Pharma companies are continually evaluating ways to address supply chain challenges to meet customer needs ultimately.
- A cloud-based agile technology network to increase visibility in the supply chain and keep all functions connected for better collaboration can boost your profits and increase growth potential.
See pharmaceutical supply chain optimization in action.
At a Glance
- The Pharmaceutical industry has witnessed exponential growth during the past decade, and pharma revenues worldwide reached approximately 1.6 trillion U.S. dollars in 2023, growing to an estimated 1.77 trillion U.S. dollars in 2024 – Per Statista.
- This growth rate automatically created a need to boost supply chain visibility and inventory traceability.
- Regulatory measures put forth by the government or agencies such as the FDA can create unforeseen challenges for Pharma companies.
- Leveraging technology’s power and identifying solutions for Pharma to streamline operations can effectively boost your bottom-line and help in better quality control while meeting both regulations and market demands together.
Understanding the Pain Points
The inability to adapt to technological advancements and rapidly changing regulatory landscape makes managing pharmaceutical supply chains quite a tough pill to swallow. Successful pharmaceutical companies leverage the power of IT to streamline their supply chain operations.
Figure 1:Pain Points in Pharmaceutical Industry

The pharma industry has established itself as one of the fastest-growing industries in the past few decades, with a significant share of its investments in research, marketing, manufacturing, and development of its products.
In the present scenario, the table seems to be turning. The industry is struggling to keep pace with rapidly changing regulations and dealing with challenges such as:
- Keeping up with the growing competition due to faster technological advances
- The rapid development of new products to handle growing health concerns.
- Healthcare providers and government reforms put downward pressure on product prices of the industry.
- Full traceability of the product pipeline to be on par with requirements like quality compliance, serialization mandate, and the rise of counterfeited drugs.
- Issues related to delivering products to the market.
The above challenges highlight the importance of comprehensive solutions that help Pharma companies progress and overcome various hurdles in shipping products to customers.
Collaboration Within the Pharma Value Chain
To keep up with the growing list of challenges, Pharma companies are increasingly relying on third parties like CROs, CMOs, 3PLs to manage the following:
- Outsource research & development.
- Manufacturing of intermediates.
- 3rd party logistics for warehouse and inventory management.
- Quality control testing.
- Shipping and distribution of products.
While this is beneficial for pharmaceutical companies, it comes with its complexities, including the remote location of different departments, inventory, and testers. How do you run a pharmaceutical supply chain effectively when all the departments are functioning from other locations? How would you effectively manage workflows that involve the exchange of information with outsourced parties on multiple tiers of your supply?
As of 2024, the United States faced an all-time high of over 300 different kinds of drug shortages in healthcare and pharmacy settings. A 2024 report showed that 62% of all drug shortages were caused by manufacturing and product quality problems resulting in supply disruptions.
Following are a few benefits for Pharma companies to be technology-centric:
- Real-time & end-to-end supply chain visibility.
- Collaboration with the Pharma value chain.
- Optimized inventories with least stock out situations.
- Higher margins through smarter channel allocations for an increasing share in the market.
- Improved customer experience.
- Visual analytics and reporting.
Digitize the Pharma Supply Chain
Companies require well-streamlined processes and collaboration using agile software solutions for Pharma that provide significant cost savings and flexibility. Technology for the Pharmaceutical industry can be a helping hand to optimize the supply chains. Advanced Solutions for Pharma increase inventory visibility and lot traceability across your supply chain, ultimately improving the overall product quality. Post COVID-19, embracing technology as a growth enabler, has become even more critical, with 89% of pharma industry leaders implementing or developing digital transformation strategies as of 2024.
Following the path of other industries like hi-tech and consumer goods, the Pharma companies also need to focus on these five initiatives:
01. Connecting and collaborating using a working business network
A digital business network sets the foundation of a multi-enterprise supply chain, where all departments are interconnected on the cloud, ensuring seamless information flow and collaboration. Unlike the outdated model based on point-to-point connections, this multi-tier network is much advanced and helps end-users stay in sync with counterparts and move quickly through the process. The ability to collaborate enables end-to-end visibility and collaboration – the two critical factors that support smooth business interactions among different business partners involved in the value chain. Seamless integration helps you get real-time visibility and coordination in your entire extended supply chain, including multiple partners.
Figure 2:Process of Temperature Control

For example, there can be immense damage caused to vaccines due to the drop in temperature in the storage areas while being transported from one place to another. Real-time data tracking can help with adequate temperature controls by notifying people when the temperatures are above or below the desired levels.
02. Estimating true demand
Demand predictions are just well-painted guesses of how much of a percentage of the population would require a particular product in the future. Most innovative pharmaceutical companies are now following demand forecasting as per consumer product companies for their OTC products. They do this by capturing massive volumes of demand-related data and feeding it into sophisticated demand planning software, which helps them predict demand effectively. The data or demand forecasts captured are shared with all supply chain partners, ensuring the pharma companies, suppliers, and CMOs align with the arrangement. Estimating actual demand helps them to bring significantly higher on-shelf availability and lowers inventory.
03. Control quality for CMOs
Pharmaceutical companies need to ensure end-to-end traceability. How do they keep track of their pharmaceutical manufacturing supply chain when a CMO is involved? Having visibility into partner manufacturing operations and tracking product quality across the multi-tier system is essential for pharma companies. Real-time visibility can be done by connecting CMOs to your Manufacturing Execution Systems. A manufacturing execution system helps capture relevant data at all production stages, providing granular visibility into plant operations. Pharma companies will effectively track material flows, lot of genealogy, processing steps, and associated parameters, such as yields or test results – critical information to ensure traceability. Quality control is of optimal importance for any serialization initiative within pharmaceutical companies.
04. Faster planning across the network
Another essential requirement for companies is to detect and respond to changes in supply and demand faster. With largely connected systems and expanding business networks, companies have a clear, end-to-end visibility of the supply chain in the Pharma company and not just in-house operations; this includes expected lead times on inventory from a contract manufacturer or raw material supplier. However, traditional planning systems can impact your decision-making capabilities required to manage inventory shortages and suggest alternative purchase/ manufacturing scenarios. Advanced planning tools allow rapid evaluation of new buy-sell signals and easily recommend purchases from secondary vendors or alternate manufacturing routes. This new plan can then be shared with all the supply chain partners within the value chain through collaboration portals, tools, and so forth.
05. Manage distribution
Pharma companies are increasingly relying on third parties for transportation, warehousing, and other value-added services. Ensuring product availability implies proper management of distribution partners. For downstream visibility, inventory management coupled with sophisticated stock strategies are needed. Proactive pharma supply chain management helps all products be delivered to the right customers to increase your sell-metric, which is critical when competing in the market.
Pharma companies are customizing supply chains to suit the product types being transported, with digitally mature pharma companies reducing development timelines by up to 30% through the implementation of real-time visibility, IoT sensors, and blockchain technology for enhanced transparency and security.
Key Takeaways
- The expectation of better customer experience is the same across all industries and progressive Lifesciences companies understand the need to embrace digital for Pharma.
- More Pharma companies are continually evaluating ways to address supply chain challenges to meet customer needs ultimately.
- A cloud-based agile technology network to increase visibility in the supply chain and keep all functions connected for better collaboration can boost your profits and increase growth potential.
See pharmaceutical supply chain optimization in action.

